Biochemistry is a big, scary word. And it does mean quite a lot of complex things. But at the end of the day, it’s this: how do the molecules that make up our cells work together to produce life?
In this blog entry we’re going to look at the most basic and important building blocks of biochemistry.
This may feel a bit like being back in science class, but it will help us to understand the general principles of biochemistry for future reference when we’re using them for the exciting task of designing alien creatures.
In this chapter, we’re going to answer the following questions:
- What are the six major chemical components necessary to build a living cell, as far as we know?
- Where can these be found in space?
- How might these chemicals differ in alien life vs. Earth life? How might they be the same?
This chapter will also provide us with groundwork for discussing life not as we know it in the chapters to come. This will allow us to intelligently speculate, alongside NASA scientists, about where in the universe life might be found outside the “habitable zone,” and what it might look like.
Now then, what are the basic ingredients for life as we know it? And why are they so ideal?
Ingredient #1: Water
Water is a lot more special than we often realize. For us, it’s just about the most common liquid in nature. It’s the basis of pretty much everything we drink, and of all our bodily fluids. Alien life could use any other liquid instead of water. Right?
Well, it turns out water, not so much. Other liquids might not be able to facilitate biochemical reactions in the way liquid water does.
Why is that? It’s because of water’s molecular structure.
You’ve probably heard that the chemical formula for water is H2O. Two hydrogen molecules, one oxygen molecule. What you might not have realized is that this is a very special combination in terms of chemical reactivity.
Hydrogen isn’t very good at hanging onto electrons. In fact, it sucks at it. This is due to a few principles that will be useful to know before we move on:
Charged Particles Want to Be Neutralized
All atoms are made up of positively charged protons and negatively charged electrons. This is important because these oppositely-charged particles “want” to be paired together.
Charged particles will stick to particles with the opposite charge, in an effort to neutralize their own charges. This is one of the major forces that keeps atoms together. But there are also other forces at play, and the tug-of-war between the attraction of protons and electrons to each other + these other principles creates most of biochemistry.
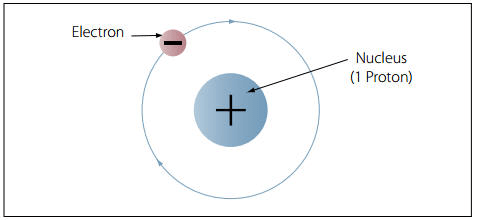
Electrons Want to be Paired
Electrons don’t just want to stick to a proton that can neutralize its charge. They also “want” to be paired up with another electron.
Atoms that manage to pair up all their electrons and balance their positive and negative charges are very stable, and they don’t much “want” to react with other chemicals. They’re happy just the way they are.
Examples of such substances are helium and the other “noble gases” – so called because they snobbishly refuse to interact with other chemicals. These elements that are very happy all by themselves tend not to show up in biochemistry too often, because biochemistry requires that atoms want something from each other. This is what allows them to form the chemical bonds that makes complex organic molecules, and life as we know it, possible.
In water molecules, the electrons are “happy” because they’re paired. By sharing electrons with each oxygen, hydrogen can give each of its electrons a buddy.
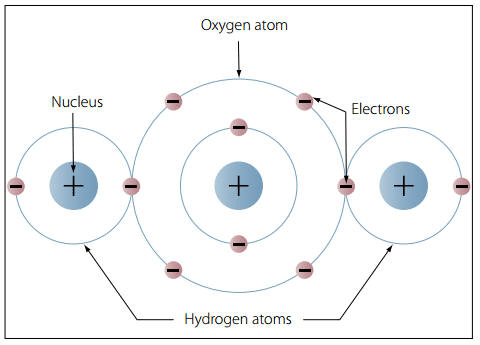
Hydrogen and Oxygen Are Clingy When They Get Together
Now, both of the components of water – hydrogen and oxygen – are pretty unhappy types of atoms.
Hydrogen has only one proton and one electron. That electron desperately wants another electron buddy, and its proton often isn’t strong enough to hang onto the atom’s electron. For that reason, electrons belonging to hydrogen atoms often go wandering, attracted to the stronger electrical charge of larger atoms’ nuclei.
This can also cause hydrogen atoms’ own proton nuclei to develop full to partial positive charges, which means they start grabbing other atoms’ electrons to replace their own.
Now, oxygen is a very greedy chemical partner. It, too, isn’t happy with the number of electrons it has – but it is one of the most “electronegative” elements on the periodic table, meaning that an oxygen atom’s nucleus attracts electrons very strongly.
This means that when one oxygen and two hydrogen atoms get together, the arrangement of electrons is lopsided. The electrons gravitate disproportionately toward the oxygen atom’s big, powerful nucleus, and the poor little lonely hydrogen atom nuclei are left with partial positive charges.
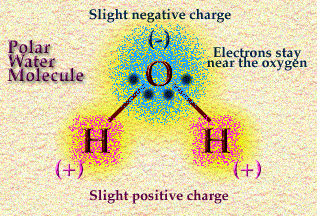
This turns out to give water a bunch of special properties, which make it an ideal solvent for the biochemistry of life as we know it.
Water is stable enough that it doesn’t just destroy other molecules submerged in it. The partial charges of the oxygen and hydrogen atoms balance each other out. But those partial charges can also encourage chemical interactions, by gently tugging on the protons and electrons of other molecules.
Scientists call this phenomenon “hydrogen bonding.” This refers to the bonds that form between separate water molecules. A lonely hydrogen atom in one molecule, for example, might cozy up to the nucleus of a different water molecule’s oxygen atom in order to be close to the oxygen’s electrons.
This means that water molecules don’t just have chemical bonds isolated within their own molecular structure – they also form unusually strong bonds with other molecules, and tug on their protons and electrons. Water can do this with other water molecules – but it can also do it to nucleic acids, amino acids, and other essential molecules for life.
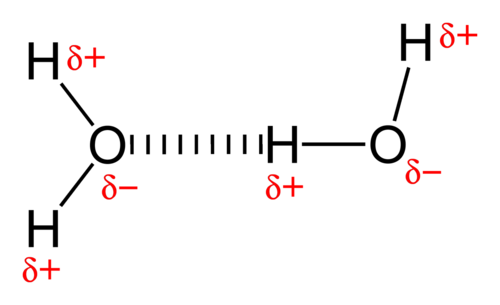
This allows water to dissolve nearly anything – that is, to pick up other molecules and hold them in solution by tugging on their protons and electrons. This is why you might notice that oil or rubbing alcohol won’t dissolve certain substances – but water will.
And it means that if you mix other chemicals together in a solution of water, they’re more likely to react with each other, too. This is because their protons and electrons are already being loosened up by their clingy friends, the water molecules. Think of it like a party where the clingy hydrogen and oxygen molecules are always peer pressuring other chemicals to interact with each other.
This also gives water some unique benefits that can be seen even outside of biochemical reactions.
Capillary action, for example – the principle that causes water to run uphill to soak a paper towel, or run all the way to a top of a tree to feed the tree’s leaves – is caused by water’s clinginess. The partial charges of the hydrogen and oxygen atoms try to bond with the atoms of your paper towel, or the tree’s capillaries – and this process actually pulls water up the capillaries against the force of gravity.
Water is also the only liquid we know of that expands instead of contracting when it freezes. This, too, may be vital to life.
It turns out that most liquids get more dense when they freeze, rather than less dense. The only reason water doesn’t is that its molecules form a lattice according to hydrogen bonding. The “desire” of one molecule’s hydrogen atoms to be close to the oxygen atom of a different molecule forms a latticework of molecules, and actually forces the molecules further apart.
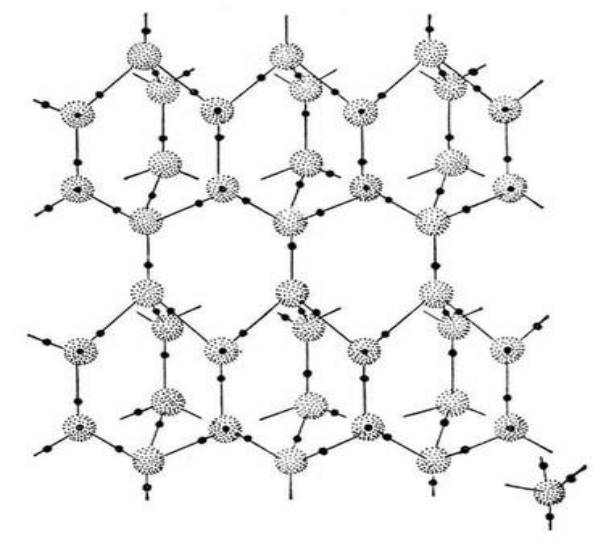
If this didn’t happen, frozen water would sink to the cold bottom of oceans and lakes instead of floating on top. This could rapidly lead to all the oceans and lakes on Earth being frozen solid in a self-sustaining cycle. Instead, when water freezes, it becomes less dense and floats to the surface – which allows sunlight and currents of warm water which also rise to the top of bodies of water to melt it.
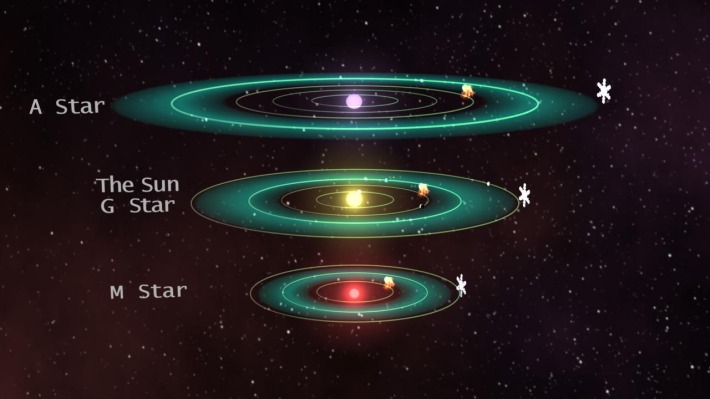
This is all why NASA scientists define the “habitable zone” around a star as being “the zone in which liquid water could exist on the planet’s surface.” As far as we know, most other liquids just won’t be able to do the job of keeping the oceans from freezing solid, and encouraging other vital biological molecules to react with each other in the ways that allow life as we know it to exist.
So what other kinds of chemicals are necessary for living cells? And how does water help them interact with each other in the necessary ways?
We’ll cover that in our next few blogs, which will examine how some essential chemical components make life possible – and how these might be adjusted in alien life to suit the needs of your plot.

This is a beautiful reading. I love this! Thank you very much for this information. 🥰
LikeLike