On Earth, the most common forms of energy storage molecules are sugars. These are rings made of oxygen, carbon, and hydrogen atoms which store chemical energy in their bonds.
How exactly does this work, you ask? Well, chemical bonds take energy to create. They also release energy when they are broken. The more chemical bonds a molecule has – provided your cells have the necessary enzymes to break those bonds into a form of chemical energy they can use – the more energy it can store.
Sugars don’t store as much energy as fat molecules, but they’re easier to make and break down. For this reason, they’re the energy storage form most frequently used by plants. The starch found in grains, edible roots, and fruits are made of sugar molecules daisy-chained together.
In the case of the plant parts I’ve mentioned above, the sugars are actually intended as energy storage to sustain the plant through a cold, sunless winter (in the case of roots), or to sustain baby plants before they can grow their own leaves (in the case of fruits and grains).
How do plants store the Sun’s light in the form of chemical energy? They have organelles called chloroplasts, which contain the green pigment chlorophyll. Chlorophyll absorbs certain wavelengths of light – literally absorbing their energy, and using it to knock electrons loose from the pigment molecule. These high-energy electrons are then used to create high-energy bonds, like the carbon-oxygen bonds in sugars.
Below you can see a chemical diagram of a molecule of common baking sugar, in all its glory. Just imagine all the chemical energy taken from the Sun to power your metabolism tied up in those sweet, sweet carbon-carbon and carbon-oxygen bonds:
What’s important to know here is this: living things can find ways of turning almost any form of heat or light energy into chemical energy, and storing it. These storage molecules allow living things to keep living for a while, even if they have to shed their leaves or wander away from their geothermal vent.
The Chirality of Sugars
Just like amino acids and nucleic acids, sugars have “handedness.” They can exist as mirror images which have identical chemical properties, but which can’t be digested by the same enzymes.
In our case, life on Earth uses almost exclusively “right-handed” sugars. We don’t know if there’s a good reason for this, so alien life could use “left-handed” sugars – sugars which show up as identical to our own on some chemical tests, but which we would not be able to digest or use for energy.
This raises the possibility of interesting plot points. The colonists who arrive on a lush alien planet, only to realize they have no ability to digest the types of sugars and proteins used by its life forms. The colonists who are sure they will contract a terrible alien plague – only to find out at the last minute that the bacteria can’t digest their sugars and so can’t survive inside their bodies.
There are also much, much more exotic ways to design alien metabolisms, such as…
Acetylene, Titan, and Life Not As We Know It
One particularly exciting hobby for astrobiologists studying other planets is looking for chemicals that could be byproducts of this type of metabolism. Oxygen is actually a byproduct of photosynthesis: when plants stick some carbon and oxygen molecules together to make a sugar, two oxygen atoms are left over, and these molecularly bond to each other.
Oxygen gas, conveniently, can then be used to break the sugars plants make back down into chemical energy by us. When we break the carbon and oxygen chemical bonds they’ve created and use the energy stored in those bonds to power our own metabolism, we exhale the component parts of the sugars – carbon dioxide.
Fats are another type of energy storage molecule that can store even more energy for longer periods of time, because it’s just backed full of carbon-hydrogen bonds. Fat molecules store more than twice as much energy by weight as sugar molecules. But, there’s a catch.
Fat molecules are less stable than sugar molecules over time, so life forms that don’t need to move around tend to favor sugars as a fuel source. Only complex animals, who need to prioritize mobility over long-term stability, benefit from using fat.
So when astrobiologists look for molecules that could be used as alien energy sources among the stars, they look for molecules that are similar to sugars, or similar to the byproducts of making sugars. This principle of looking for molecules that could come from energy storage by biological life has led to some exciting findings.
On Mars, scientists have noticed that methane concentrations in the atmosphere are higher during warmer parts of the year. While this could be caused by geological outgassing, some scientists believe it might instead be caused by bacterial life. On Earth, some species of bacteria produce methane as their “byproduct” of turning sunlight or heat into energy storage molecules. On Mars, Martian bacteria could be doing the same.
On Saturn’s moon Titan, on the other hand, scientists have been intrigued by some unexpected findings. Although Titan is too cold for life as we know it, some have proposed it as a good model for life not as we know it, which we’ll discuss in a later chapter. And one tantalizing sign of this? There’s less acetylene – an organic molecule with chemical bonds similar to sugar – than there should be on Titan’s surface.
Acetylene may look very different from a sugar molecule, but scientists say that its triple-bond between carbon molecules could be broken by living things to yield energy in the same way as sugar’s carbon-oxygen and carbon-carbon bonds.
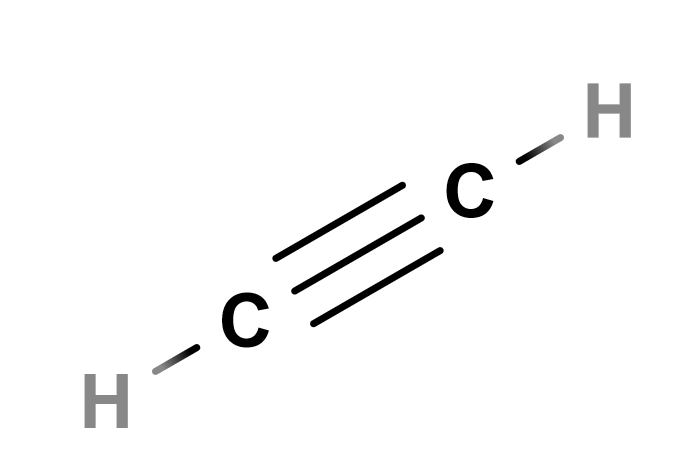
Scientists have calculated Titan’s expected surface and atmospheric composition, based on those of nearby planets and moons. They expected to find more hydrogen and more acetylene, but when they examine Titan’s surface in detail, they find these chemicals in shorter supply than expected.
Now, we know that Titan has liquid on its surface – liquid methane, ethane, and propane, existing in unimaginably cold and relatively sunless conditions. But the sugar-like molecule acetylene is made by natural processes on Titan, and its similarity to sugar has caused some scientists to ask:
If it’s disappearing, could something be eating it?
Life forms on Earth use molecules similar to acetylene for fuel, and they can also inhale hydrogen to drive their metabolic processes. Could a shortage of hydrogen and acetylene on Titan suggest that this moon hosts life not as we know it?
No one knows yet, and it could be many years before we can get a probe with the sufficient analytical capabilities to land safely on Titan’s incredibly cold surface. But some scientists think that we could find methane-based life – an exciting new type of life that would expand our definition of a star’s “habitable zone” – when we do finally get there.

I’ve saved the discussion of Titan’s biosphere for last in this blog sequence because it’s an excellent intro to the possibilities of life not as we know it.
We’ll be covering more alien star and planet types, and more alternative biochemistries and metabolisms available for use in alien life, throughout the summer. If you haven’t gotten a chance to do so yet, you can sign up to follow my blog, or sign up for my newsletter to be notified when my whole big ol’ alien worldbuilding book becomes available.

